Determination of Ammonia in Water Samples by Phenate Method
Course Information
Subject: ENCE 305 – Sanitary Engineering
Year/Part: III Year I Part
Description: Lab report covering Determination of Ammonia in Water Samples by Phenate Method and Spectrophotometric Analysis

Determination of Ammonia in Water Samples by Phenate Method
1. Objective
To determine the concentration of Ammonium Nitrogen (NH4–N) in given water samples (Drinking Water and Non-Drinking Water) using the Phenate Method and spectrophotometric analysis.
2. Equipment and Apparatus
- Glassware: Volumetric flasks (500 ml, 100 ml, 50 ml), Transfer pipettes, Pipettes (10 ml, 5 ml, 1 ml), Screw bottles (30 ml), 100 ml Beaker, Test tubes with caps.
- Instrumentation: Analytical balance (Chemical balance), Spectrophotometer, Glass cells (1 cm).
- Filtration: Syringe, Syringe filter (0.2 µm cellulose acetate).
- Others: Pure/Distilled water bottles.
3. Required Chemicals and Reagents
3.1 Chemicals
- Distilled water
- Phenol (G.R.)
- Sodium Nitroprusside (G.R.)
- Sodium Hydroxide (NaOH)
- Sodium Hypochlorite (5% or 12%)
- Ammonium Nitrogen Standard Solution (1000 mg-NH4–N/L)
3.2 Reagents Preparation
- Solution A (Phenol/Sodium Nitroprusside): To approximately 400 ml of water, 5 g of Phenol and 0.025 g of Sodium Nitroprusside were added and diluted to 500 ml. This solution was prepared one day prior to analysis to ensure complete dissolution and stored in a dark bottle.
- Solution B (Sodium Hypochlorite): Commercial sodium hypochlorite solution and 7.5 g of Sodium Hydroxide were dissolved in 100 ml of water and diluted to 500 ml to achieve an effective chlorine concentration of approximately 0.1% w/v. The solution was stored in a dark bottle and refrigerated.
4. Theory
4.1 Definition
Ammonia (NH3) is a colourless gas with a pungent odour. In water, it exists in equilibrium between un-ionized ammonia (NH3) and ammonium ions (NH4+). It is a common pollutant produced by the hydrolysis of urea and the biological degradation of organic nitrogenous matter.
4.2 Principle
The Phenate Method relies on the reaction of ammonia, hypochlorite, and phenol catalyzed by sodium nitroprusside to form an intense blue compound known as Indophenol. The intensity of the developed blue colour is directly proportional to the concentration of ammonia in the sample. According to Beer-Lambert’s Law, the absorbance of the solution measured at 640 nm is directly proportional to the concentration of ammonium nitrogen (NH4–N) present in the sample.
4.3 Guideline Values
While there is no specific health-based guideline value for ammonia in drinking water by WHO, it is an indicator of possible bacterial, sewage, or animal waste pollution. The taste and odour threshold for ammonia is approximately 1.5 mg/L. Natural levels in groundwater and surface water are typically below 0.2 mg/L.
5. Procedure
5.1 Preparation of Standard Solutions (Weight Method)
- The chemical balance was powered on and allowed to complete its calibration.
- A screw bottle was placed on the balance, the cover was closed, and the “tare” button was pressed to zero the reading.
- The required amount of “Stock Solution” or “Standard Solution A” (Std. Sol. A) was weighed out carefully, and the cover was closed.
- The actual weight of the solution was recorded.
- Pure water was added to the bottle until the total weight reached 25 g.
- The actual gross weight was recorded.
- The screw bottle was capped and mixed well.
- The actual concentration was calculated using the formula:
5.2 Ammonia Analysis by Phenate Method
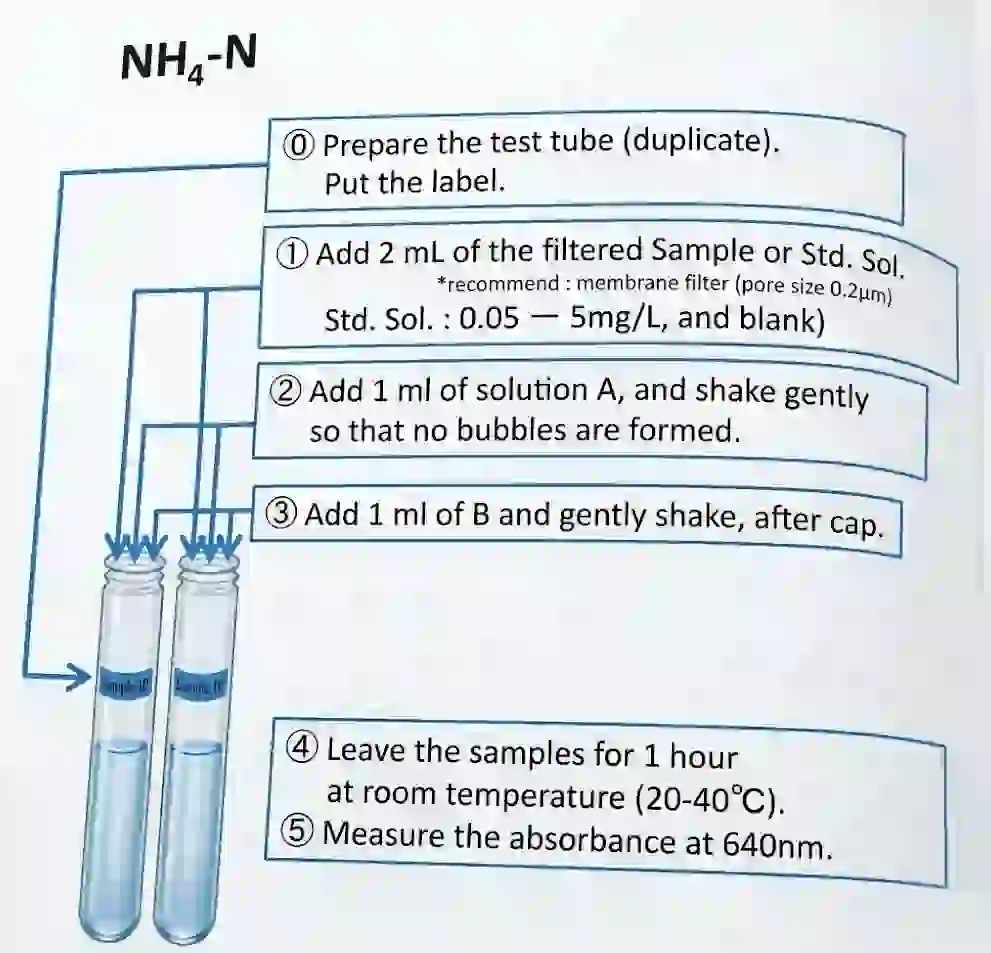
- Removal of suspended particles: The sample was filtered through a 0.2 µm syringe filter.
-
Sample Preparation:
- All samples were prepared in duplicate.
- 2 ml of the sample was taken in a tube (dilution was performed if necessary to bring the final volume to 2 ml).
- 1 ml of Solution A was added to all samples, blanks, and standard solutions. The tubes were shaken gently to avoid bubble formation.
- 1 ml of Solution B was added, and the tubes were capped and shaken gently.
- Colour Development: The samples were left for 1 hour at room temperature (20–40°C) to allow the bluish-green colour to develop.
- Measurement: The absorbance was measured at 640 nm using a spectrophotometer.
6. Observation
6.1 Standard Curve Data
Table 1: Absorbance of Standard Solutions
| Concentration (NH4–N mg/L) | Absorbance (at 640 nm) |
|---|---|
| 0.0 (Blank) | 0.000 |
| 0.1 | 0.016 |
| 0.2 | 0.094 |
| 1.0 | 0.101 |
| 3.0 | 1.265 |
| 5.0 | 1.827 |
6.2 Sample Data
Table 2: Absorbance of Water Samples
| Sample ID | Absorbance (Observed) |
|---|---|
| Drinking Water | 0.189 |
| Non-Drinking Water | 3.000 |
7. Calculations and Graph
7.1 Standard Calibration Curve
A linear regression analysis was performed on the standard data. The equation of the line is given by y = mx + c, where y is Absorbance and x is Concentration.
7.2 Sample Calculation
Using the regression equation: Absorbance = 0.389 × Concentration − 0.063
Rearranging for Concentration (x):
1. Drinking Water (y = 0.189):
2. Non-Drinking Water (y = 3.000):
8. Result
The concentration of Ammonium Nitrogen (NH4–N) in the samples was found to be:
Drinking Water: 0.65 mg/L
Non-Drinking Water: 7.87 mg/L (Extrapolated)
9. Discussion
The calibration curve shows a positive correlation between concentration and absorbance, although some non-linearity was observed in the mid-range data points (specifically the 1.0 mg/L standard). The Drinking Water sample falls within the calibration range and is below the odour threshold of 1.5 mg/L. The Non-Drinking Water sample showed an extremely high absorbance (3.0), indicating a very high concentration of ammonia, significantly exceeding the calibration range. For accurate analysis of the Non-Drinking Water, the sample should be diluted and re-analyzed to bring the absorbance within the linear range (0.0 – 1.0 Abs).
10. Engineering Significance
Ammonia nitrogen is a critical parameter in sanitary engineering:
- Pollution Indicator: High levels indicate fresh contamination by sewage or agricultural runoff.
- Chlorine Demand: In water treatment, ammonia reacts with chlorine to form chloramines, increasing the chlorine dosage required to achieve break-point chlorination.
- Toxicity: High concentrations can be toxic to aquatic life, particularly fish.
- Corrosion: It can cause corrosion in copper pipes and fittings.
11. Precautions
- Chemical Safety: Phenol is toxic and can cause burns; gloves were worn during handling.
- Fresh Reagents: Solution A was prepared a day in advance to ensure stability, while Solution B was kept refrigerated.
- Contamination: All glassware was thoroughly rinsed with ammonia-free water to prevent cross-contamination.
- Bubble Removal: Samples were mixed gently to prevent air bubbles, which can interfere with spectrophotometric readings.
Disclaimer
The educational materials provided on this website are intended as supplementary resources to support your learning journey. These lab materials are sample documents designed to help students understand sanitary engineering lab techniques.
We have made every effort to ensure the accuracy of the content. However, we recommend students to refer to standard lab manuals and consult with professors for authoritative explanations. These materials should be used as references only.
We respect intellectual property rights. If you believe any content should be credited differently or removed, please don’t hesitate to contact us. We’re happy to make appropriate corrections or give proper attribution.
